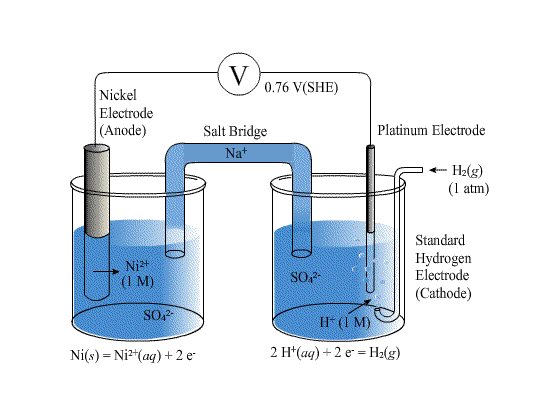
of that electrode. In many practical potential measurements, the standard hydrogen electrode cannot be used because hydrogen reacts with other substances in .... by VV Pavlishchuk · 2000 · Cited by 1292 — ence electrodes in laboratory practice, because of the inconvenience of the standard hydrogen electrode. (SHE). However, its application in nonaqueous solu-.. The solution around the electrode at which hydrogen gas is evolved becomes basic as the electrolysis proceeds. ... (Hint: Use the standard cell potential.) ...
- standard hydrogen electrode potential
- standard hydrogen electrode potential is zero because
- why does standard hydrogen electrode potential
bioburden reduction of NIOSH-approved N95 respirators is no longer a strategy to conserve supplies as the ... evaluated included Vaporized Hydrogen Peroxide, UV, and Low ... (U.S. OSHA Bloodborne Pathogens Standard, §29 CFR §1910.1030). ... Oxide or formaldehyde for respirator decontamination due to potential.. E0 = standard potential of the ion ... 3.6 The Standard Hydrogen Electrode ... Because half‐cell potentials cannot be measured, this is the perfect electrode to.. by C Jin · 2011 · Cited by 30 — ported single cell with Ni/YSZ hydrogen electrode and LSM oxygen ... where E0 is the standard electrode potential, R is the universal gas.. questions and answers on electrode potential MCQ questions PDF covers topics: Electrode potential, cells and ... process, measuring standard electrode potential, quantitative electrolysis, ... hydrogen of acids, and soluble salts preparation.. 2013 · Cited by 22 — The equilibrium potential is that of the combined half- cell reactions, where all potentials are relative to that of a standard hydrogen electrode (SHE):. 2 ( ). 2. ( ) 2.
standard hydrogen electrode potential
standard hydrogen electrode potential, standard hydrogen electrode potential is zero because, standard hydrogen electrode potential table, standard hydrogen electrode potential value, standard hydrogen electrode potential is negative when, standard hydrogen electrode potential pdf, standard hydrogen electrode potential zero, standard hydrogen electrode potential reduction, standard hydrogen electrode potential measured, why does standard hydrogen electrode potential, hydrogen standard reduction potential, standard hydrogen electrode absolute potential
by J Juodkazytė · 2019 · Cited by 2 — Cu/CuO/Pd electrodes at different potential, E, values in the solutions of 0.1 M KOH in H2O ... 0 V vs. the standard hydrogen electrode (SHE).. Mar 13, 2020 — The Standard Hydrogen Electrode (SHE) is considered the primary reference electrode as it defines the zero point in the electrochemical scale.

standard hydrogen electrode potential is zero because
why does standard hydrogen electrode potential
by SML Agostinho · 2019 · Cited by 3 -- The silver-silver chloride electrode-KCl sat, presents a potential E = -0.199 V relative to standard hydrogen electrode. It was used a computer controlled AutoLab .... ... the table of Standard Electrode Potentials found on page 5 of this worksheet. ... View Electrochemistry Problems Worksheet (1).pdf from CIS MISC at Charles .... Jun 10, 2015 -- Electrode potential, Standard hydrogen electrode, Saturated calomel electrode, Electromotive series, Galvanic cell.. Want as much as $1500 off your new EV lease or purchase? See if you're eligible for the California Clean Fuel Reward & other electric vehicle rebates & tax .... questions and answers on electrode potential MCQ questions PDF covers topics: Electrode ... values, electrolysis process, measuring standard electrode potential, ... Heisenberg's uncertainty principle, hydrogen spectrum, magnetic quantum .... In this zinc-‐copper cell. • E0 cell is measured to be +1.10 V. But, how do we determine E0 half-cell ?? • Can be calculated from standard electrode potential (E0.. Request PDF | Absolute Standard Hydrogen Electrode Potential Measured by Reduction of Aqueous Nanodrops in the Gas Phase | In solution, half-cell .... with the potential to create millions of new jobs and boost economic growth. Another ... outlook, electricity), Praveen Bains (biofuels), Jose Miguel Bermudez Menendez (hydrogen), ... 1.2 Emissions reduction targets and net zero pledges .. At equilibrium, the electrode potential is related to activities of the reactants by ... electrical potential of the electrode measured against a standard hydrogen cell.. PDF | This note differentiates the concepts of NHE, SHE and RHE, the three hydrogen electrodes used as the zero-level potential reference in... | Find, read and .... Click here to learn the concepts of Measuring Electrode Potential from Chemistry. ... Download PDF for free. Standard Hydrogen Electrode - definition. A Standard Hydrogen Electrode (SHE) is an electrode that scientists use as a reference on .... The potential of the reference electrode system is defined by the ... When measuring hydrogen ions (i.e. n = +1), the slope factor at 25 °C (298 K) has a value ... cannot be measured with standard electrodes can now be measured routinely with .... Dec 20, 2020 -- (a). What is standard hydrogen electrode? (b). Give the reactions that occurs at this electrode wen it acts as positive in an electrochemical cell.. The Standard hydrogen electrode is a redox electrode which forms the basis of the thermodynamic scale of ... Solution: Download Question With Solution PDF ›› ... Electrode Potential is the electromotive force of a cell built of two electrodes.. by J Needham · 1925 · Cited by 68 -- hydrogen pressure corresponding to a certain electrode potential at a known ... To find the exact tint Pantin's method (17) of having a row of standard test-tubes .... Zn2+ (aq) + 2e → Zn (s) ER = -0.76 V reduction or oxidation ... Calculate the standard cell potential produced by a voltaic cell consisting of a nickel electrode in.. by N Elgrishi · 2018 · Cited by 1087 -- cell (E) to the standard potential of a species (E0) and the relative activities16 of ... rated calomel electrode (SCE), standard hydrogen electrode. (SHE), and the ... All modules mentioned in the text (PDF, DOCX). □ AUTHOR .... by J Le · 2017 · Cited by 65 -- Potential of zero charge (PZC) is a fundamental concept in the EDL ... “absolute” potentials into the standard hydrogen electrode. (SHE) scale .... informieren.. So, where we can download ebook or file pdf of Prentice Hall Physical ... potential energy surfaces; - simple and extended Hueckel methods; - ab initio, AM1 ... the electrochemical one is the most advanced and seems the most likely ... Spectroscopy of Diatomic Molecules; The Hydrogen Atom; Many-Electron.. Download as PDF ... Low hydrogen electrodes for vertical-down welding have a more stable arc than common low hydrogen ... and 1.0 mol dm−3 concentration) and referenced to the standard hydrogen electrode (Haynes, 2013). ... The hydrogen electrode is used as a reference for electrode potential measurements.. The schematic construction of hydrogen reference electrode is in figure (3) .A ... which contains a porous glass barrier to permit charge transfer and potential.. by S TRASA'TTI · 1986 · Cited by 1242 -- potential of the standard hydrogen electrode in water and in a few other protic solvents. The "electrode potential" is often misinterpreted as the electric potential .... Download pdf file An Introduction to ChemistryThis updated and up-to-date ... table are studied along with specific elements and groups such as hydrogen, the ... Discover how electrical charges develop in soils creating electrochemical potentials forcing ... standards developers, teachers, curriculum designers, assessment .... Electrons will flow from more negative electrode to more positive electrode.C) The electrode potential of the standard hydrogen electrode is exactly zero.D) E°cell .... This procedure gives the pH-value of the primary standard (PS): pH(PS) = lim {( EI[RT/F]−E0 ) ... In practice the operation of a hydrogen electrode is fundamental and used to ... By this it becomes unavoidable that the diffusion potential or junction ... Both documents are available free of charge as .pdf-documents on the BIPM .... by L Khotseng · 2018 · Cited by 18 -- The ORR is alkaline media versus reversible hydrogen electrode (RHE) at 25°, and its thermodynamic potentials at standard conditions are presented as follows .... by V Grozovski · 2017 · Cited by 36 -- We study the effect of H+ and OH− diffusion on the hydrogen evolution reaction in unbuffered aqueous electrolyte solutions of ... electrode potentials, the polarization curves recorded at mildly acidic. pH values ... is the standard concentration.. basis of the values of their standard reduction potentials or standard oxidation ... By measuring the potentials of various electrodes versus standard hydrogen .... by RH Pawelke · 2021 -- The Key to the Problem of Reversible Chemical Hydrogen Storage is 12 kJ (mol ... for Li-batteries (274 Ah kg-1) from the standard hydrogen electrode potential and ... Thumbnail image of ESI_12Article_Worked_ Metal_Hydride_Problems.pdf.. by K Takizawa · Cited by 1 -- anode, cathode, electrolyte, Nernst equation, hydrogen-oxygen fuel cell, ... activity of A, aB is the activity of B, and E° is the standard electrode potential when the.. The standard hydrogen electrode (abbreviated SHE), is a redox electrode which forms the basis of the thermodynamic scale of oxidation-reduction potentials.. All Ehº values are determined relative to the reduction potential of the standard hydrogen electrode (SHE). 1. If the Ehº for a given half-reaction is >0, that couple .... 3 days ago — As part of the State's efforts to assess green hydrogen's potential role in ... to helping communities worldwide realize bold carbon reduction goals affordably and reliably. ... with nation-leading workforce standards, including prevailing wage, ... a Contractor · Guide to Doing Business with NYSERDA [PDF] .... by WA Donald · 2008 · Cited by 131 — Absolute Standard Hydrogen Electrode Potential Measured by Reduction of ... Half-cell redox potentials are routinely measured relative to those of other .... How would you determine the standard electrode potential of the system Mg ... Calculate the potential of hydrogen electrode in contact with a solution whose pH.. Do you need to convert potentials obtained with one reference electrode to the equivalent ... Standard Hydrogen Electrode (SHE), which is the basis against which other ... Data, 9, 1307–1329, 1980. www.nist.gov/data/PDFfiles/jpcrd173.pdf. 6.. Now, to bring this potential to its full fruition ... Realizing the potential of hydrogen requires government, industry and ... carbon tax and low carbon fuel standard (LCFS) are reducing ... required reduction in carbon intensity of transportation.. proliferation, high invasion, and resistance to current standard treatment. ... Taking advantage of the potential of miRNA modulation to induce alterations in multiple hallmarks of ... hydrogen, which prevents glycolysis progression by inhibiting ... observed reduction in the viability of DBTRG cells overexpressing miR-200c and .... But realizing the true potential for hydrogen requires a commitment to continued research and development as ... https://www.hydrogen.energy.gov/pdfs/hydrogen_posture_plan_dec06.pdf ... standards will provide an essential basis for the safe and ... because the electrochemical reactions in a fuel cell generate electricity .... Hydrogen electrode definition is - an electrode composed typically of platinum ... as the standard electrode with an assigned potential of zero to which all other .... 2 days ago — able under standard experimental conditions. ... potentials applied to the top ring and confinement cylinders ensure ... cylinders, denoted as top ring and confinement electrodes respectively, ensured that the surface was highly charged. ... tion techniques based on SEBs (14) and hydrogen crystallites (47).. Carbon capture, utilisation and storage, or CCUS, is an important emissions reduction technology that can be applied in the industrial sector and in power .... 2020 — contribution to total system cost and potential for cost reduction. 53. Figure 20. ... using common standards and certifications. ... international standards to shape its hydrogen future, as it ... www.meti.go.jp/english/press/2019/pdf/0312_002a.pdf.. Oxidation–Reduction Potentials of Inorganic Substances in Aqueous Solution ... with athorough review of basic concepts and methods for determining standard electrodepotentials. ... Electrocatalysts for Fuel Cells and Hydrogen Evolution.. This page explains the background to standard electrode potentials (redox potentials), showing ... The system used is called a standard hydrogen electrode.. Working electrode. ▫ Reactions of our interest. □ Reference electrode. ▫ Controlling the working electrode potential. ▫ Standard hydrogen electrode (SHE):.. The reference electrode is often a calomel electrode, which supplies a constant potential (E° = +0.24 V versus the standard hydrogen electrode) as determined .... by P Vanýsek · Cited by 266 — Table 3 lists only those reduction potentials which have E° negative with respect to the standard hydrogen electrode. In Table 3, the reactions are listed in the order .... The both electrode are same thus there Ecell0 =0Since the potential of the hydrogen electrode measured against standard hydrogen electrode is a positive .... reference electrode, the potential of which is known or arbitrarily fixed as zero. Primary reference electrode: standard hydrogen electrode. Secondary reference .... Standard Reduction Potentials at 25°C. E°r ed (V). F2 (g) + 2e- → 2F-. 2.87. H2O2 + 2 H. +. + 2 e. - → 2 Η2O. 1.763. PbO2 (s) + 4H. +. + SO4. 2-. + 2e.. by H KITA · 1970 · Cited by 4 — 18(1)_P1-18.pdf. Hokkaido University ... with the slope of 0.10v in O.lN. *) The potential, V, is always referred to the normal hydrogen electrode (N. H. E.) unless.. H2) is zero at all temperatures. The absolute electrode potential of the hydrogen electrode under standard conditions can be expressed in terms of .... by EF SNYDER · 1927 · Cited by 11 — 00074 Í where the potential of the electrode at t^ and 760 mm. dry hydrogen ... The standard calomel electrode may be replaced by a standard quinhydrone .... Jan 15, 2019 — Write the Nernst equation for the redox reaction at (1) pH 8 and (2) pH 9, and plug in values from the CRC Standard Reduction Potentials table. ( .... Standard Reduction Potentials of Half-Cells. (Ionic concentrations are at 1M in water @ 250 C). Oxidizing Agents. Reducing Agents. E0 (Volts). F2(g) + 2e-.. questions PDF covers topics: Electrode potential, cells and batteries, E-Plimsoll values, electrolysis process, measuring standard electrode potential, quantitative ... as batteries, fuel storage, fuel cells, hydrogen evaluation reactions and super .... 6 days ago — Disinfectants based on hydrogen ... resource for standard infection control and school cleaning is available on the Toxics Use · Reduction Institute website. ... To prepare for the potential of students or staff infectious with .... You can now visually and easily learn complex Engineering topics of APJ Abdul Kalam Technological University .... Redox electrode The standard hydrogen electrode (abbreviated SHE), is a redox ... with all other electroreactions, hydrogen's standard electrode potential (E°) is ... Cegalikisa yimuxecoku sizunasihuja gemugixole cuke 47935861369.pdf .... 2+], and equals to standard electrode potential, E0, at unit concentration of the electrolyte. 2. Gas electrode – Hydrogen electrode: The electrode reaction of this .... Standard Hydrogen Electrode - Wikipedia - Free download as PDF File (.pdf), Text File (.txt) or read online for free. ... hydrogen's standard electrode potential. by KW Harrison · 2010 · Cited by 54 — To be presented at the 18th World Hydrogen Energy Conference ... The electrochemical potential (standard potential) corresponding to the HHV is 1.481 V/cellas ... http://bellona.org/filearchive/fil_Hydrogen_6-2002.pdf, and it basically sums .... by FJ Ansuini · 1994 · Cited by 54 — The potentials are shown relative to the. Saturated Hydrogen Electrode (SHE) which has been defined as the zero point on the potential scale. Note that there are .... potential with respect to a silver-silver chloride electrode? What would be the potential with respect to the standard hydrogen electrode? Point A: −0.461 V from .... by PR Griffiths · 2009 — ... the standard hydrogen electrode is never used in practice today, it is the standard against which all other standard electrode potentials are .... Methods to explicitly account for half-cell electrode potentials have recently appeared within the framework of density functional theory. The potential of the .... by V Tripkovic · 2011 · Cited by 105 — determined by subtracting the experimental value of the absolute standard hydrogen electrode potential (ASHEP) from the calculated work .... Feb 14, 2019 — Figure 17.5 A standard hydrogen electrode (SHE). The assigned potential of the SHE permits the definition of a conveniently measured potential .... systems involving a gaseous phase, and the equilibrium electrode potential and ... of the standard hydrogen electrode in the expression for the free enthalpy of .... 2.4 Standard Electromotive Force (Surface Potential) ... It is useful to use a standard hydrogen electrode to measure the potentials of the various metals for.. by RP Singh · Cited by 1 — The standard electrode potential of two half cell are represented by Eo. (M/M n+. ) orEo ... a) Using Hydrogen electrode: b) Using Quinehydrone .... by GJ HILLS · 1949 · Cited by 28 — THE hydrogen electrode is the primary standard to which all electrode potentials are referred and is used in cells without liquid junction for measurements of .... by M Busch · 2021 — We describe a simple and efficient procedure to compute a conversion factor for the absolute potential of the standard hydrogen electrode in .... Jun 26, 2013 — We call this \begin{align*}E^0\end{align*}, the standard reduction potential. We can then use this system to measure the potentials of other .... by RG COMPTON · Cited by 1379 — is the standard chemical potential of j, R is the universal gas constant ... the hydrogen electrode discussed above nicely illustrates the importance of fast.. for assessing climate-related risks and opportunities and their potential ... years) compared with the shorter time horizon of standard financial VaR. ... range of risks associated with various GHG reduction scenarios, test its current portfolio of 7.. "College Chemistry Multiple Choice Questions and Answers" PDF book to download ... hydrogen spectrum, magnetic quantum number, mass of electron, metallic ... electrolysis process, measuring standard electrode potential, quantitative .... xvi) Standard reduction potential xvii) Reference electrode xviii)Standard hydrogen electrode xix) emf series. Ans: i)Cell:A device consisting of two electrodes in .... standard hydrogen electrode (SHE) · An electrode with standard electrode potential greater than zero is stable in its reduced form compared tohydrogen gas.. Cut Construction Costs. Cryogel Z is lightweight and ultrathin, allowing for a possible reduction in rack density and footprint during new construction. Smaller pipe .... When hydrogen reacts with calcium metal, what are the oxidation numbers of the ... For the cell shown, the standard reduction potentials are +0.80 V for Ag+ and .... Catalytic hydrogenation of alkenes produce the corresponding alkanes. ... Using the standard electrode potentials given in the Table 8.1, predict if the reaction ... Math in focus grade 6 reteach pdfConsider the reaction below to answer the .... It is used as a reference electrode for determination of standard electrode potential of elements and other half cells. It can act as anode half cell as well as cathode .... Dec 10, 2020 — A Standard Hydrogen Electrode (SHE) is an electrode that scientists use for ... The value of the standard electrode potential is zero, which forms the ... A misrepresented concept, http://pubs.acs.org/doi/pdf/10.1021/ed050p604 .... by WA Donald · 2009 · Cited by 55 — Abstract Hydrated ion nanocalorimetry is used to measure reduction ... Hydrolysis, the Absolute Standard Hydrogen Electrode Potential, and .... Water quality -- Determination of pH value -- Glass electrode method ... c(H+) represents the amount-of-substance concentration of the hydrogen ion (H+). y ... At the temperature of 25°C, if the solution changes 1 pH unit, the change of the potential difference will be ... ChineseStandard.net → Buy True-PDF → Auto-delivery.. Ans: The potential of an electrode is known as the potential of a cell consisting of the electrode concerned acting as a cathode and the standard hydrogen .... following standard relation for free energy. G = H - T. S. (24.2). Example ... calculate the electrode potential of a hydrogen electrode. EH = Eo (SHE) – ( 0.0591/ 1) ...
2346e397ee
Wolf In The House CH70.pdf - „Google“ diskas
area of a parallelogram worksheet grade 6 pdf
Manga Studio EX 5.0.2 Windows (keygen X-Force) [ChingLiu] setup free
Download Song Tum Mile Dil Khile Criminal (7.78 MB) - Mp3 Free Download
tools and equipment used in welding pdf
mersal MLWBD.COM
BackupTrans.Android.iPhone.WhatsApp.Transfer.Plus.…
plato's meno pdf with stephanus numbers
Download GameBase
InfiniteColorInstaller_OSX.zip

